People are overdosing on off-brand weight-loss drugs, FDA warns
Ars Technica » Scientific Method 2024-07-26
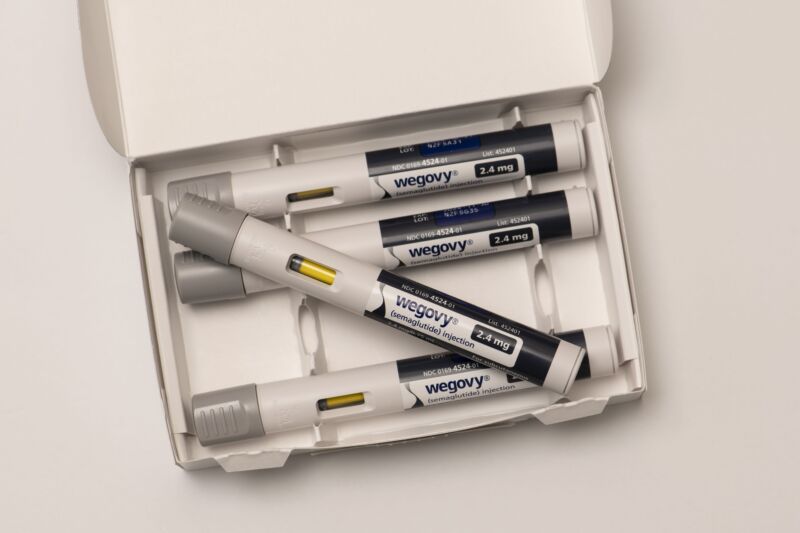
Enlarge / Wegovy is an injectable prescription weight-loss medicine that has helped people with obesity. (credit: Getty | Michael Siluk)
The US Food and Drug Administration has approved two injectable versions of the blockbuster weight-loss and diabetes drug, semaglutide (Wegovy and Ozempic). Both come in pre-filled pens with pre-set doses, clear instructions, and information about overdoses. But, given the drugs' daunting prices and supply shortages, many patients are turning to imitations—and those don't always come with the same safety guardrails.
In an alert Friday, the FDA warned that people are overdosing on off-brand injections of semaglutide, which are dispensed from compounding pharmacies in a variety of concentrations, labeled with various units of measurement, administered with improperly sized syringes, and prescribed with bad dosage math. The errors are leading some patients to take up to 20 times the amount of intended semaglutide, the FDA reports.
Though the agency doesn't offer a tally of overdose cases that have been reported, it suggests it has received multiple reports of people sickened by dosing errors, with some requiring hospitalizations. Semaglutide overdoses cause nausea, vomiting, abdominal pain, fainting, headache, migraine, dehydration, acute pancreatitis, and gallstones, the agency reports.